Sterile Disposable Plasma Apheresis Set
Short Description:
Product Detail
Product Tags
1. Device Name:Sterile Disposable Plasma Apheresis Set
2.Intended Use
The sterile disposable plasma apheresis set is used in conjunction with automated plasma collection system for the collection of human blood plasma and transfusing the red blood cell back to the blood donor.
3.Structural composition
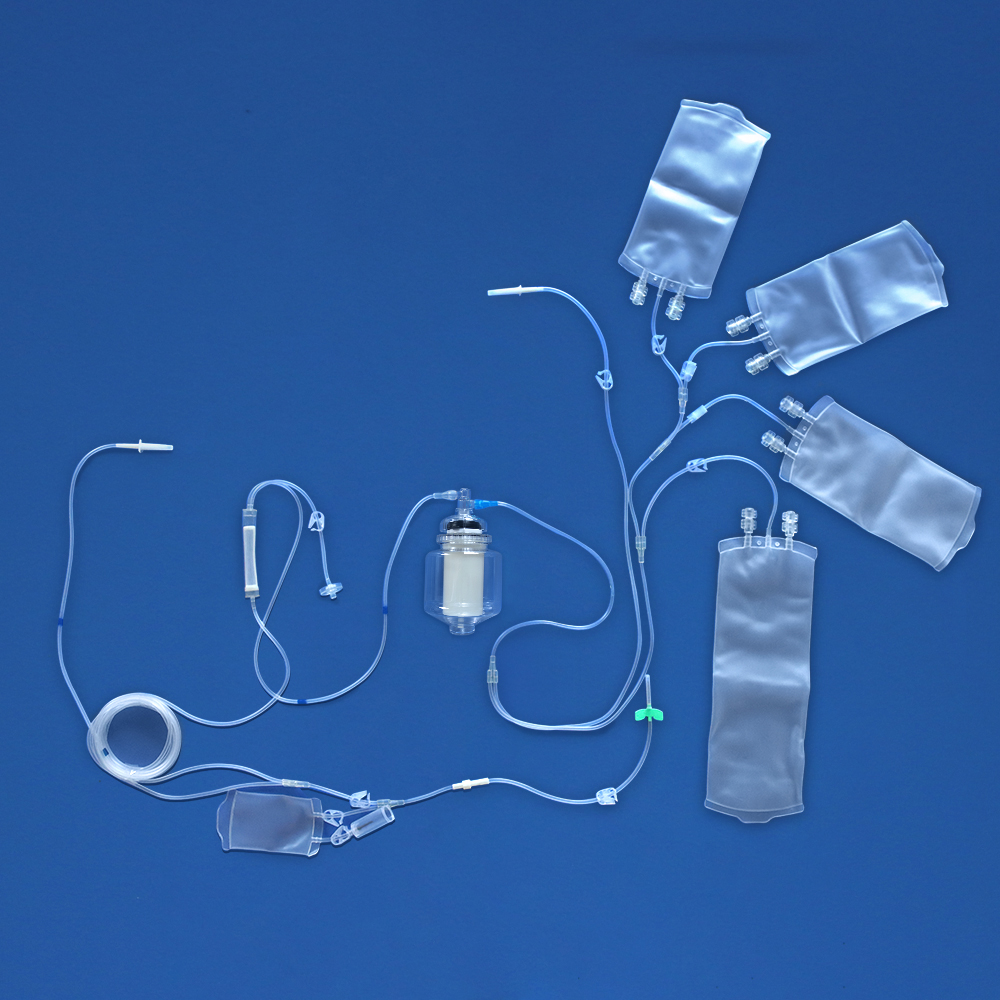
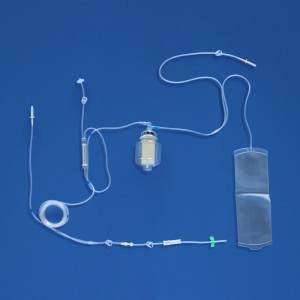
a) The sterile disposable plasma apheresis set consists of plasma pheresis cup, plasma tubing, plasma bag, and A. V. fistula needle sets, all or part of which are composed.
b) The plasma pheresis cup is mainly composed of cup, inner sheath, central tube, lower petticoat, upper petticoat, ceramic seal ring, gasket, rubber bowl, static head, protective sleeve, etc. Plasma pheresis cup can be centrifuged.
c) The plasma tubing is mainly composed of all or part of pump tube, puncture device, puncture device protective sleeve, blood and blood component filter, pressure monitor connector, separation cup connector, limit card, clamp, blood collection and transportation tube, anticoagulant tube, outer cone connector, outer cone connector protective sleeve, U tee joint, outer cone tee, tee protective sleeve, sampling bag, sampler, etc.
d) The plasma bag is mainly composed of all or part of the Cup Joint, Blood Collection & Transfusion Tube, bag (or multi bag), clamp, sampling tube, Transfusion port, U Tee joint, Y Tee joint, sampler, air filter, Closure-piercing device, Protective Cap for Closure-piercing Device, etc.
e) The A. V. fistula needle sets is mainly composed of Blood-taking needle, Sheath of the blood-taking needle, needle handle, clamp, Blood taking tube, inner cone joint, U Tee joint, sampler, sampling bag, etc.
4.Storage
The product shall be stored in a dry and ventilated place, away from corrosive gases. And the packing box shall be identified with words, like fragile, keeping away from wet and heat, and limits of stacking, etc. The storage temperature is recommended to be within 0~35℃.